|
For an irreversible expansion, this is what we define as "isothermal. Note that, for the irreversible path, the system temperature matches the surroundings temperature (isothermal) only at the interface with the surroundings, but not throughout the interior of the system. This will give you the change in entropy of the system for the irreversible path as well as for the reversible path.įor your problem, this procedure will give you the equation that you have written. (Entropy is a function of state).Ĭalculate the integral of dq/T for the reversible path that you have devised. This reversible path does not have to bear any resemblance whatsoever to the real irreversible path, other than it must pass through the same initial and final end points. Note: If you havent already read the page about introducing entropy, you should do so before you go on. Focus only on the initial equilibrium state of the system and the final equilibrium state.ĭevise a reversible path for the system that takes it from the initial equilibrium state to the final equilibrium state. calculating entropy changes This page looks at how you can calculate entropy changes during reactions from given values of entropy for each of the substances taking part. For example, you wouldnt calculate nutrition in the same way you. 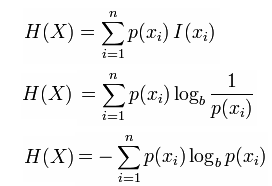
Totally forget about the irreversible path. The standard molar entropy of a substance is the absolute entropy of 1 mole of the substance in the standard state. Exactly how you calculate entropy is very field specific. This is the quantity that he called entropy, and it is represented by H in the following formula: H p1 logs(1/p1) + p2 logs(1/p2) + + pk logs(1/pk). 
Use the first law of thermodynamics to determine the final thermodynamic equilibrium state of the system for the irreversible path. The GLCM (corresponding to the pixel to the right) of the image above is computed as follows: glcm np.squeeze (greycomatrix (img, distances 1, angles 0, symmetricTrue, normedTrue)) And finally we apply this formula to calculate the entropy: where p (i, j) represents the entries of the GLCM. Solution Recognizing that this is an isothermal process, we can use Equation 5.4.1 S n R ln ( V 2 V 1) ( 1.00 m o l) ( 8.314 J / ( m o l K)) ln ( 44.8 L 22.4 L) 5. 
Also, it is concluded that if the enthalpy decreases, a reaction is successful. Calculate the entropy change for 1.00 mol of an ideal gas expanding isothermally from a volume of 24.4 L to 48.8 L. Change in Enthalpy The change of enthalpy in a reaction is almost equivalent to the energy gained or lost during a reaction. When calculating the Residual Entropy, I know that we have to use Boltzmann Equation DeltaSKb ln(w), Where Kb is a constant (1.38062 X 10-23) and where W. Here are the steps to determining the change in entropy for an irreversible process on a closed system: E U+ PV Where E is enthalpy, U is the internal energy of any system, P is pressure, and V is volume.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
